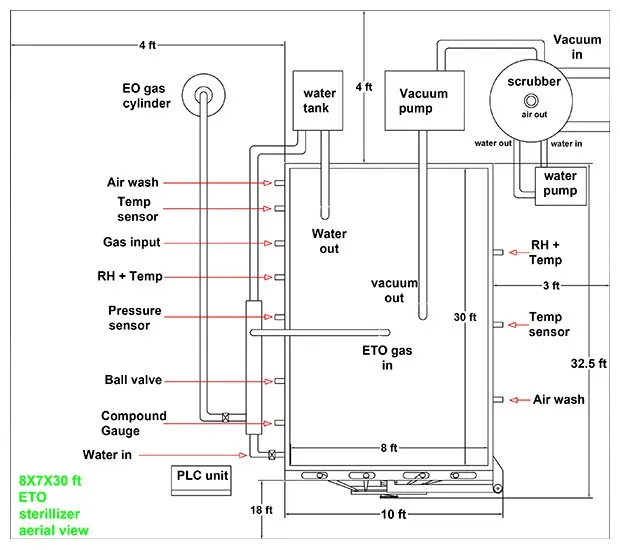
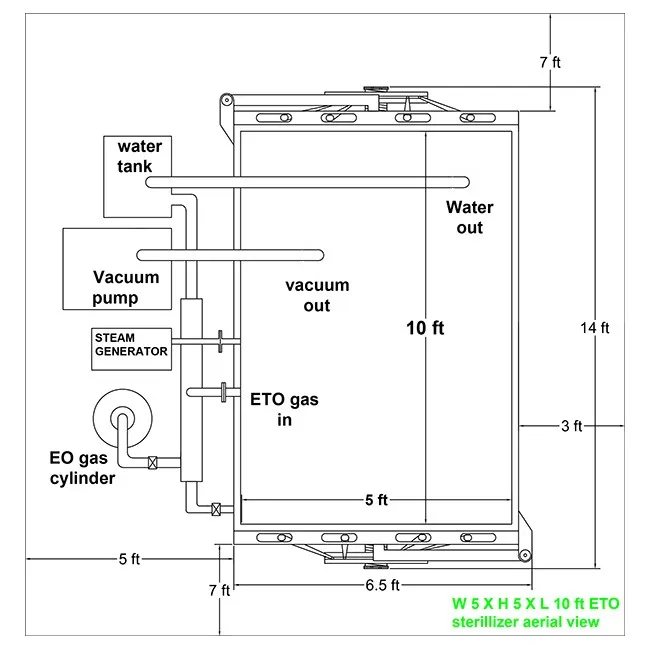
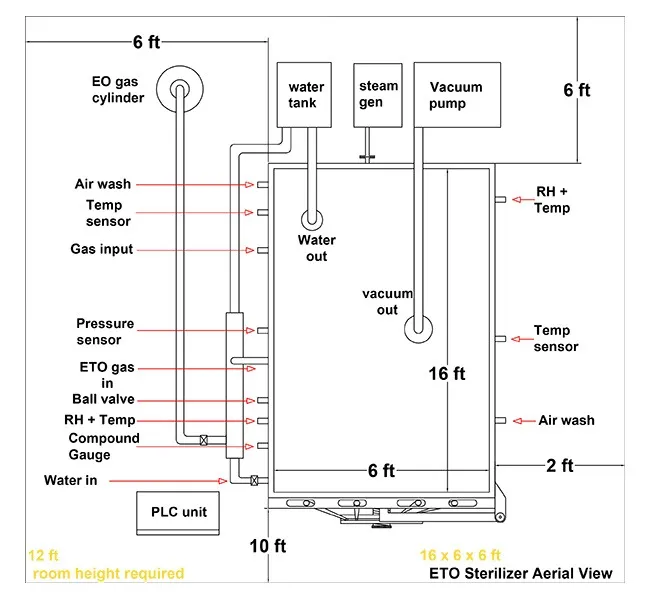
EO or EtO stands for ethylene oxide sterilization. ETO Sterilization is a chemical process that possesses critical elements and alkylation properties which cause genetic disruption in bacteria, further preventing them from producing cells. This sterilization is mostly used to disinfect medical, food, and pharmaceutical goods. As micro-organisms cannot be treated with high temperature steam disinfection and they keep on replicating, thanks to Krishna Engineering for manufacturing such varied sterilizers for the Pharmaceutical and Medical Industry and develop low temperature ETO Sterilizers with necessary industry norms and maintaining national and international guidelines.
Offered ETO Sterilizer Machine cleans tiny germ particles with the help of ethylene oxide and those particles cannot be seen with normal eyes but are harmful for human and the environment so EtO sterilization process is applied on delicate products like syringes, latex gloves, catheters, etc.




Type Of Sterilizer |
Ethylene Oxide Sterilizer |
Brand |
Krishna Engineering |
Material |
Stainless Steel |
Automation Grade |
Fully Automatic |
Warranty |
2 Year |
Chamber Dimension |
4x4x6 Feet |
Chamber Volume |
2.72 m3 |
Working Pressure |
1.0 to1.5 kg/cm2 |
Shape |
Rectangular |
Usage |
Bacterial Treatment |
Chamber Volume |
3000 L |
Insulation Wall |
Triple Wall |
Temperature Range |
45 Degree C |





We Krishna engineering made the machines from high-grade Stainless steel and mild steel as per the requirement of the client the machine is made with the proper technology and machines are manufactured with standard manufacturing process with all the required precaution and inspection.
Machine's working process